October 12, 2022
Taking Inspiration from Nature to Make Molecules Like Nature Does
Most people prefer to use one hand when signing, combing their hair, or using scissors. However, the choice of left or right hand is not a feature that exists solely in humans. Many molecules, especially organic molecules, also exhibit handedness.
In certain fields of chemistry, particularly in drug production, the handedness of molecules is crucial. For example, the drug thalidomide became notorious in the mid-20th century because its so-called right-handed version could relieve morning sickness, while the left-handed version caused severe birth defects. (We will briefly explain what chemical chirality, also known as handedness, is.)
Given the importance of this approach in chemistry, significant efforts have been made to control whether chemical synthesis produces left-handed or right-handed molecules. Now, a paper published in the journal Science by Dr. José A. Nelson, a chemistry professor at San Francisco City University, describes a new technique developed in his lab that allows for precise selection of molecular handedness during chemical reactions.
First, we need to explain the significance of handedness in molecules. To illustrate, put your hands out in front of you with your palms facing outward. The hands are mirror images of each other, right? Now, flip your left hand over. One palm is facing you and the other palm is facing away, with your thumbs and little fingers aligned in the same direction, but the hands are not identical. One hand’s fingers are curved towards you, while the other’s fingers are curved away. No matter how you twist or rotate either hand, they will always be mirror images of each other, not identical.
Some molecules are similar, a characteristic known in chemistry as chirality (from the Greek word for hand, cheir). For reasons not entirely understood, organisms tend to use one or the other form of molecules for specific purposes. For example, most sugars are right-handed, while most amino acids are left-handed.
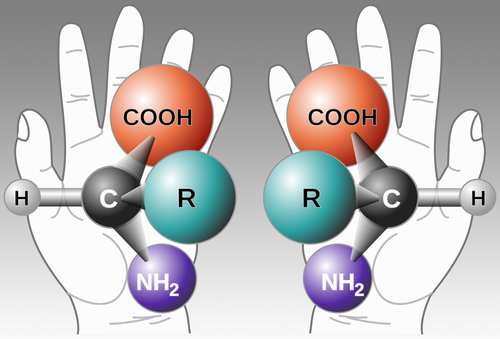
An ordinary amino acid appears in both left-handed and right-handed forms.
Nelson describes a new chemical synthesis technique in his paper, which offers high selectivity in producing either left-handed or right-handed molecules, depending on the desired substance. This technique utilizes a class of molecules known as carbocations, which are simply ions with a positively charged carbon atom.
Carbocations have a long history in chemistry due to their strong reactivity. Carbocation reactions have been used to produce polymers, steroids, and drugs, but until now, researchers have been unable to control the reaction in a way that leads to the production of only left-handed or right-handed molecules.
However, carbocation reactions can also be found throughout nature, where they are used to produce essential proteins, sugars, and many other molecules for every living organism.
Nelson said, "Carbon is a fundamental building block of all molecules in life. Nature always uses it to produce molecules of one hand or the other. We haven't been able to do that until now."
But now we can. Nelson used a class of catalysts developed by German chemist Benjamin List (who won the Nobel Prize in Chemistry in 2021) that allows for the generation of only the desired left-handed or right-handed form of a molecule.
These carbocation reactions are achieved by inserting molecular fragments containing a single carbon atom and hydrogen atoms into precise locations within larger molecules.
These reactions themselves produce the two forms of the desired molecule. The synthetic catalysts used by Nelson borrow a trick from their natural counterparts—enzymes—that forces the production of one form or the other. Enzymes and the catalysts inspired by them work through a reaction chamber, where the shape of the chamber determines which form will be created.
Nelson hopes to see this reaction become more practically applicable, moving from the laboratory environment to the pharmaceutical chemistry field, and eventually to be used for more efficient drug synthesis.
